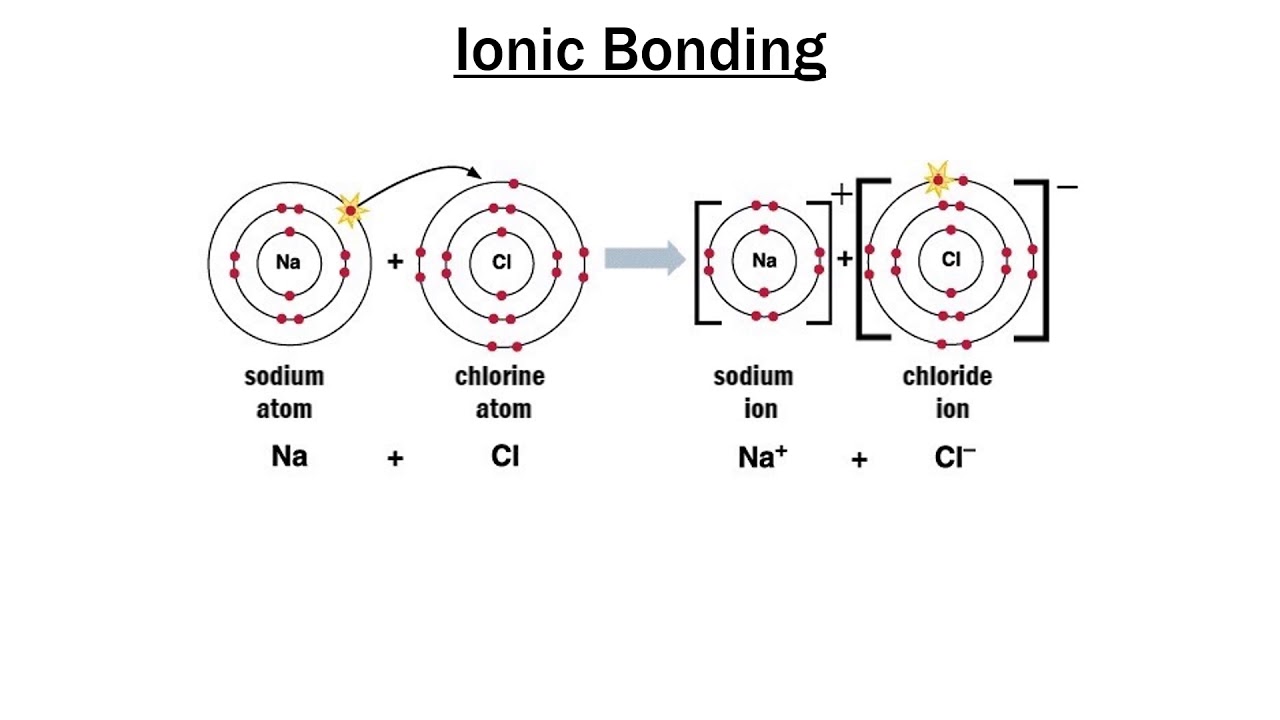
the M shell then the L shell becomes the outermost shell and it has a stable octet. There is only one electron in the outermost shell of Sodium (Na) atom and if it loses that electron from its outermost shell i.e. In comparison to molecular compounds, ionic compounds have higher enthalpies of fusion and vaporization.Ionic compounds do not conduct electricity in a solid-state but they do conduct electricity in the molten state.Electrovalent compounds usually dissolve in water and are insoluble in solvents like oil, petrol, kerosene, etc.These compounds are brittle and break into small pieces easily.The electrostatic forces of attraction between oppositely charged ions lead to the formation of ions.Ionic Compounds have high boiling and melting points as they’re very strong and require a lot of energy to break.Learn more about Physical Properties of Metal and Non-Metals here in detail. Chemical Properties of Metals and Nonmetals.Physical Properties of Metals and Nonmetals.Browse more Topics under Metals And Nonmetals Iron Oxide (Fe 2O 3) also known as rust is also an ionic bond, it has metallic compound Iron (Fe) which contains positively charged cations and it has nonmetallic compound Oxygen (O) which contains negatively charged anions. Here, Magnesium is a metal with cations and Hydroxide is a nonmetal with anions. Another example of an ionic bond is Magnesium Hydroxide Mg(OH)2 which a combination of Magnesium (Mg) and Hydroxide (OH). 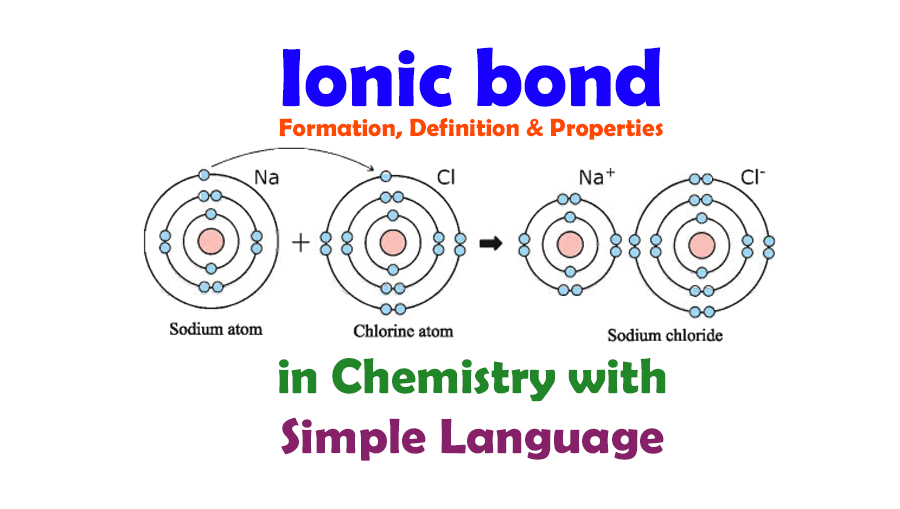
Salt (NaCl) is an ionic bond that consists of Sodium (Na) which is a metal with positive charge combines with Chlorine (Cl), a nonmetal with a negative charge. Ionic bonds are generally between metals and non-metals.įor e.g. Anions are ions that have negative charge and cations are ions that tend to have a positive charge. Nonmetals lose electrons to gain negative charge and form anions, whereas metals lose electrons to gain positive charge and form cations. Atoms that gain or lose electrons are called ions, ions may have a negative charge or positive charge. Compounds that consist of ions are known as ionic atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
